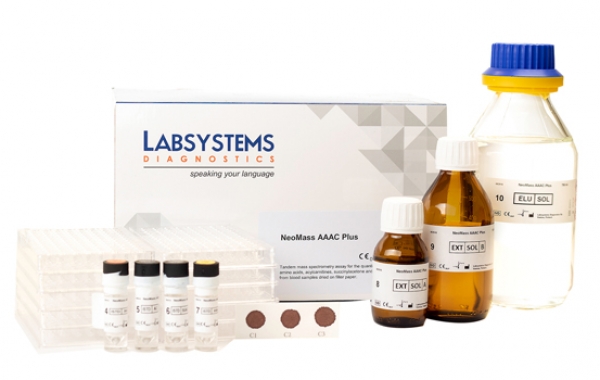
Cystic Fibrosis (CF) is an autosomal recessive disorder affecting the lungs, pancreas, intestine and liver. CF is the most common in Caucasians with an incidence rate 1:2500 newborns. CF is caused by mutation in the gene for the protein cystic fibrosis transmembrane conductance regulator (CTFR) [1,2]. Large-scale neonatal screening for CF is performed by indentification of increased levels of immunoreactive trypsinogen (IRT) in blood. Diagnosis of CF is suggested by manifestations of chronic sinopulmonary disease and exocrine pancreatic insufficiency, confirmed by a sweat test.
Neoscreen IRT EIA is a solid phase Enzyme immunoassay in which IRT is eluted from dried blood disks. It simultaneously forms a sandwich between the solid phase, coated with an antibody that recognizes Trypsinogen-1 (IRT-1) and excess of conjugate and unbound hIRT is washed away after incubation and the enzymatic reaction with Chromogenic substrate solution ( 3,3',5,5'-Tetramethylbenzidine (TMB) is performed. Read More